Naltrexone
Pharmacology and
Clinical Utility
There are a limited number of pharmacological agents currently approved for the treatment of Substance Use Disorders. Naltrexone is FDA approved and has clinical utility in the treatment of both Opioid and Alcohol use disorders.
Although not a new drug it has consistently been shown to be under prescribed for these indications. Lack of knowledge concerning prescribing information and indications has been shown to be common among practicing physicians who are often unfamiliar with basic concepts of addiction as a treatable condition. Patients are frequently unaware of this option or may be concerned about the idea of medication to treat a substance addiction.
Since its introduction additional properties and therapeutic applications for other conditions have been found. Some of these are briefly discussed in this post.

The first opioid antagonist Naloxone (Narcan) was developed in 1961 and approved for use in reversal of opioid overdose in 1971. It remains a mainstay in that role today. It is now available in a predosed nasal spray requiring minimal training to administer. A large scale public education effort by NGOs and government has resulted in measurable effects in reducing opioid overdose deaths according to recent data.
Several years later researchers at Endo labs synthesized Naltrexone a closely related longer acting opioid antagonist. The patent was later acquired by DuPont pharmaceuticals. The drug attracted little interest until DuPont was approached by the National Institute on Drug Abuse (NIDA) in 1974. The NIDA was interested in development of a non-narcotic alternative treatment for heroin addiction. Methadone was the only agent available at the time. Early studies were limited due to significant patient dropout rates with the daily oral regimen. The drug was FDA approved in 1984 for treatment of opiate dependence.
A long acting intramuscular depot injectable formulation was subsequently developed and approved for use in alcohol use disorder in 2006 and for opioid use disorder in 2010. The injection can be given once a month. This has significantly improved compliance rates and outcomes.

Naltrexone is synthesized from the reaction of oxycodone with hydrochloric acid. Oral naltrexone readily absorbs through the GI tract. It undergoes extensive first pass metabolism in the liver with subsequent bioavailability of 5-40%. The primary metabolite 6-β-Naltrexol however, is also active as an opioid antagonist.
Naltrexone binds to the three major classes of opioid receptors μ, κ, and δ. Elimination is by urinary excretion with elimination half lives of the oral drug and metabolite of 4 and 13 hours. It is generally well tolerated with few reported adverse effects. Typical oral dose is once daily. It is a pure opioid antagonist and will block therapeutic opioid doses for up to 24 hours after a single oral dose.
Opioid dependent patients may experience precipitous withdrawal if naltrexone treatment is given early. A 1-2 week gap is required prior to initiation of treatment.
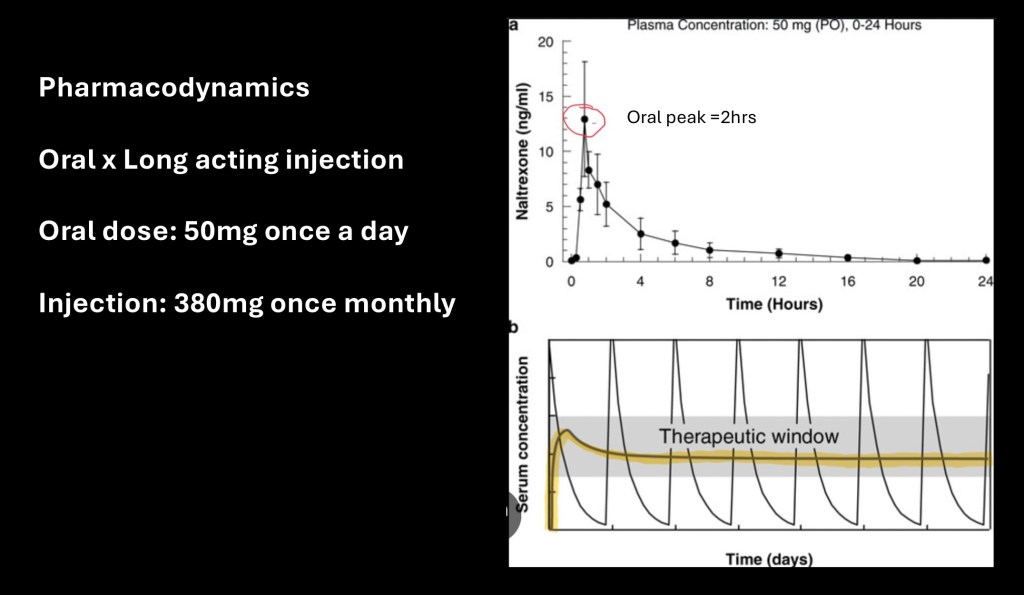
The upper graph demonstrates plasma concentration following a single 50 mg dose of oral naltrexone. There is a rapid rise to peak concentration at about 2 hours.
The lower graph superimposes daily oral naltrexone levels and a single long acting injectable dose (yellow) over a one week period. Consistent release of the medication results in a steady state plasma concentration within therapeutic range for up to 30 days.
Oral medication has been limited in effect due to high patient dropout rates. Improved abstinence rates and decreased consumption have been noted with the injectable formulation. The technology uses drug embedded within polymer microspheres. The polymer slowly dissolves due to hydrolysis releasing a steady stream of medication into the tissue. This method also bypasses first pass metabolism by the liver.
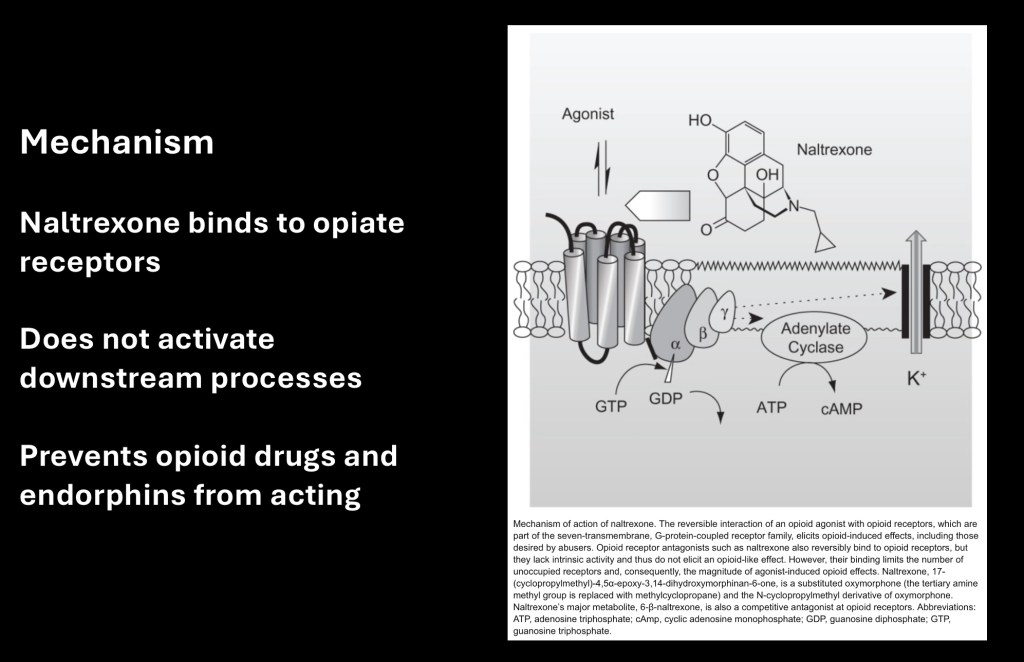
Mechanism of action is illustrated above. Opioids, either drug or endogenous endorphins bind to opiate receptors on neurons. This initiates a cascade of reactions (g-protein coupled) in the cell. This causes an influx of positively charged K+ potassium ions slowing or preventing nerve conduction.
Antagonists such as naltrexone bind to the same receptor site however they do not initiate any downstream activity within the cell. If an opiate is already present the agonist will replace it. So long as it is present administered opiates will not have any significant effect.
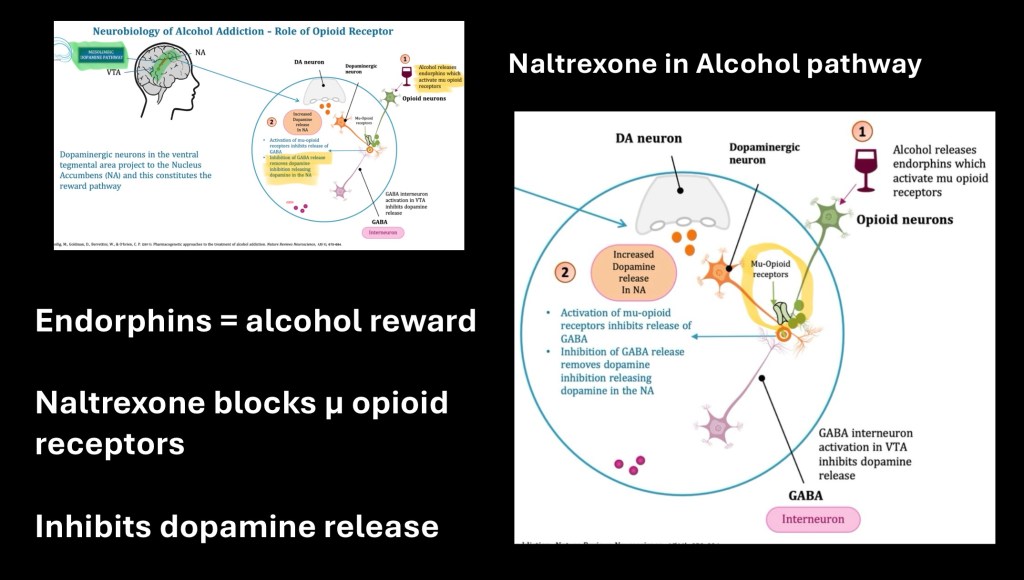
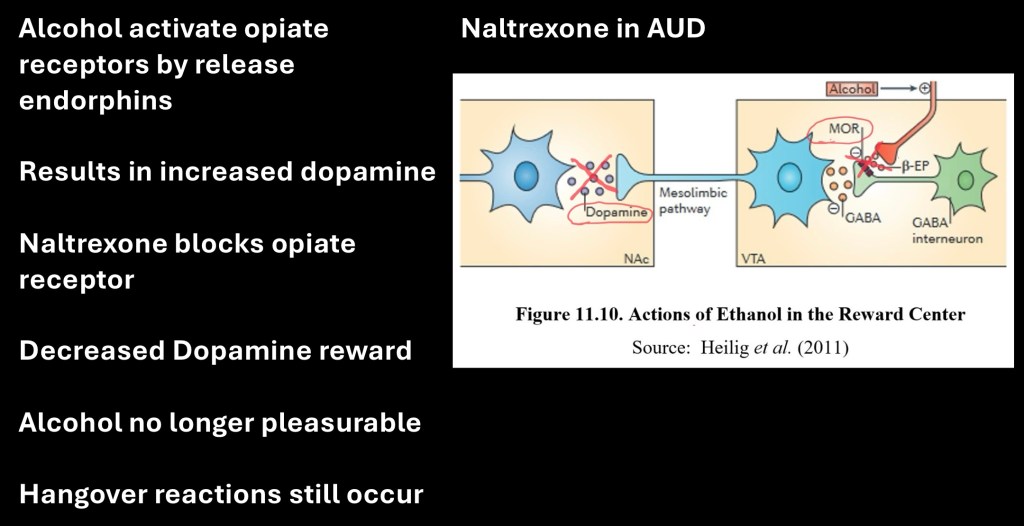
It was unexpected when when reports emerged of a decrease in alcohol use and euphoric effects of alcohol in naltrexone users. Additional studies demonstrated that endorphins are partly responsible for the rewarding effects of alcohol.
The above graphic depicts the alcohol reward mechanism
– Alcohol results in endorphin release from the hypothalamus
– β-endorphin binds to GABA receptors inhibiting GABA release
– Without GABA inhibition the dopamine neurons fire releasing dopamine and activating the reward system
By blocking the opiate receptor these actions do not occur. Dopamine is not released in response to alcohol.

Treatment for opiate dependence and withdrawal.
Opiate withdrawal can be markedly distressing and may include nausea, diarrhea, sleep disturbance, and irritability. Treatment is symptomatic. Clonidine is an α-2 sympathetic nervous system antagonist used in withdrawal.
Methadone is a synthetic long-acting opioid. It lacks the sedating functional impairment present in heroin and fentanyl. Use is restricted to approved centers and patients need to physically be present to receive doses. It is available in a liquid oral concentrate. Maintenance therapy allows users to work and other productive functions greatly improving quality of life. Discontinuance results in significant withdrawal symptoms longer in duration than heroin due to the long half life of methodone.
Buprenorphine is an opiod with partial receptor binding. It is often combined with a small amount of naltrexone (suboxone) to discourage increased dose by patients. Because of the antagonist a gap is required to initiate therapy. Dispensing is restricted but does not require a special clinic.
Naltrexone is an oral or injected opioid antagonist. It may be prescribed by any physician on an outpatient basis. It is non- narcotic. It may be used for AUD or OUD to aid in relapse prevention.
With any of these agents long term relapse prevention is improved when used along with supportive therapy/peer support group attendance.
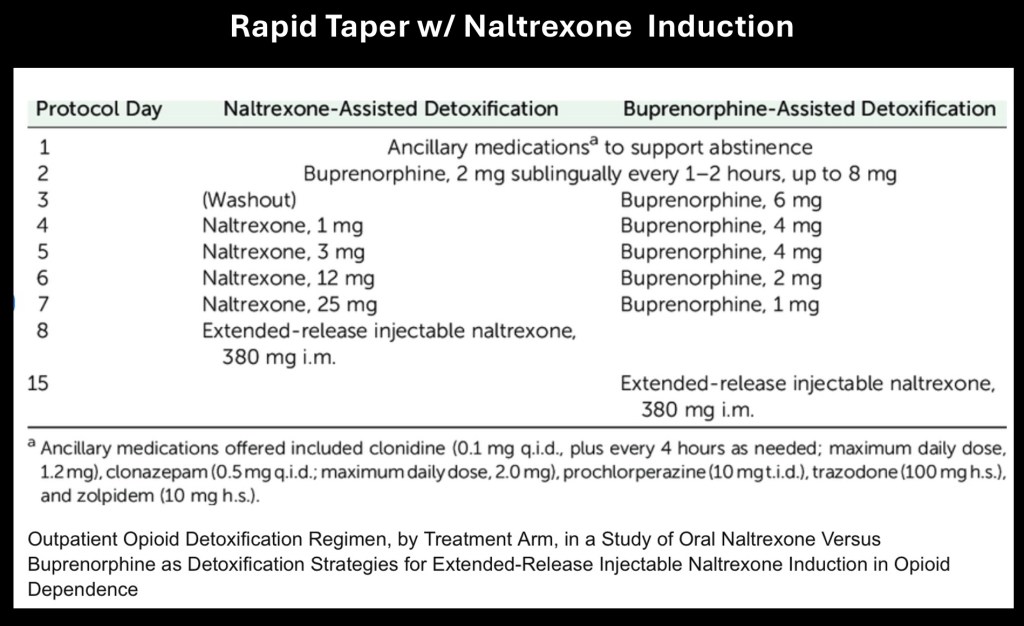
The above is an evidence based suggested schedule for faster stabilization and induction of naltrexone therapy. The schedule gradually tapers opioid dose using buprenorphine with increasing dose of oral naltrexone followed by long acting injectable naltrexone therapy. Other agents to treat withdrawal symptoms can still be used. It is designed to be administered on an outpatient basis.
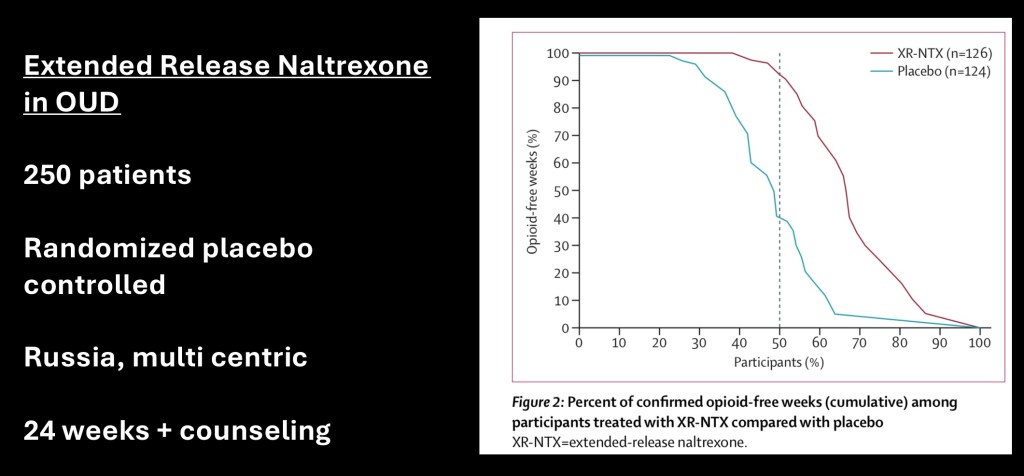
This is from a blinded placebo controlled study on injectable naltrexone performed with 250 patients. The study was multi centric and performed in Russia. The study was carried out to 24 weeks and included individual counseling. The data reflects percent of participants vs alcohol free weeks. The purple curve on the right indicates significantly more alcohol free weeks in the naltrexone group compared with placebo.
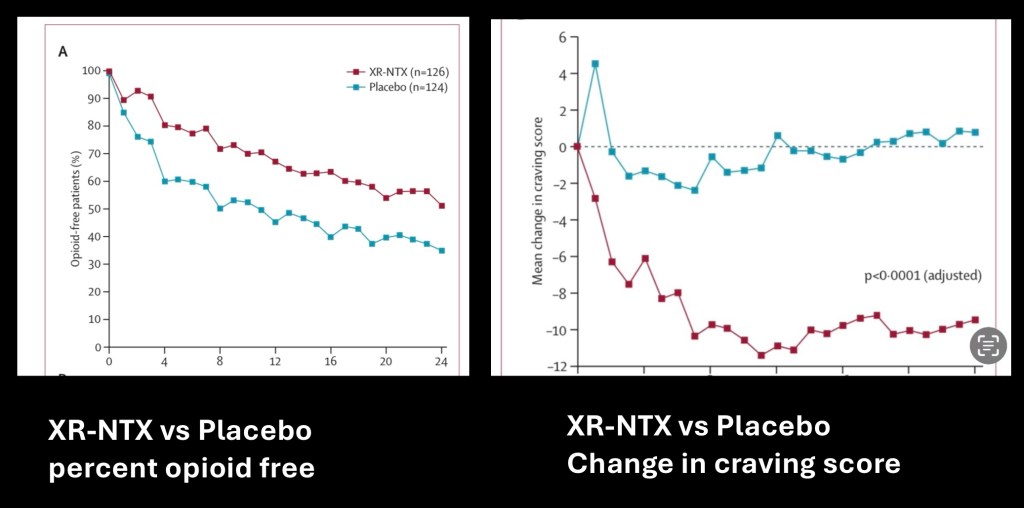
Additional data from the same study. The graph on the left shows percentage of subjects remaining opioid free over the 24 week duration of the study. Significantly more subjects remained opiate free during the study period.
The data on the right reflects change in subjective craving experienced over time compared with baseline. The naltrexone group reported significantly less craving compared with placebo.
There are three medications currently approved for treatment of alcohol use disorder by the FDA. These are Antabuse, acamprosate, and naltrexone.
Mechanism of naltrexone and the opiate system in alcohol use is depicted above. Blocking of opiate receptors results in decreased euphoria and reward with alcohol use. Post-drinking hangover symptoms may still occur when large quantities of alcohol are ingested.
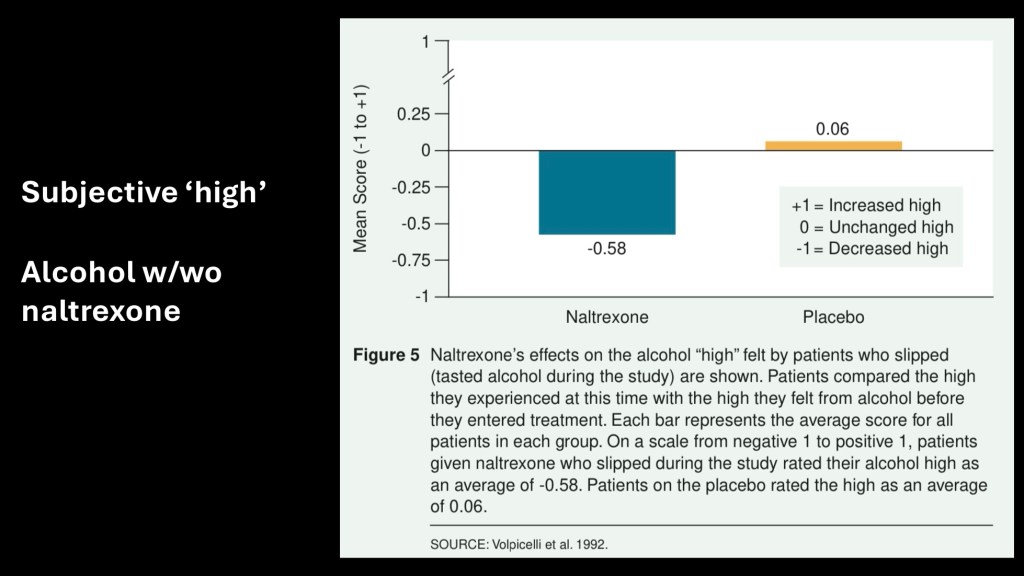
A series of studies performed in Pennsylvania by Volpicelli in 1992 was one of the earliest to explore the use of naltrexone in AUD. This data reflects the reported euphoric “high” compared with pre-treatment effect. The treated group reported significantly decreased “high” with treatment while the control group was unchanged.
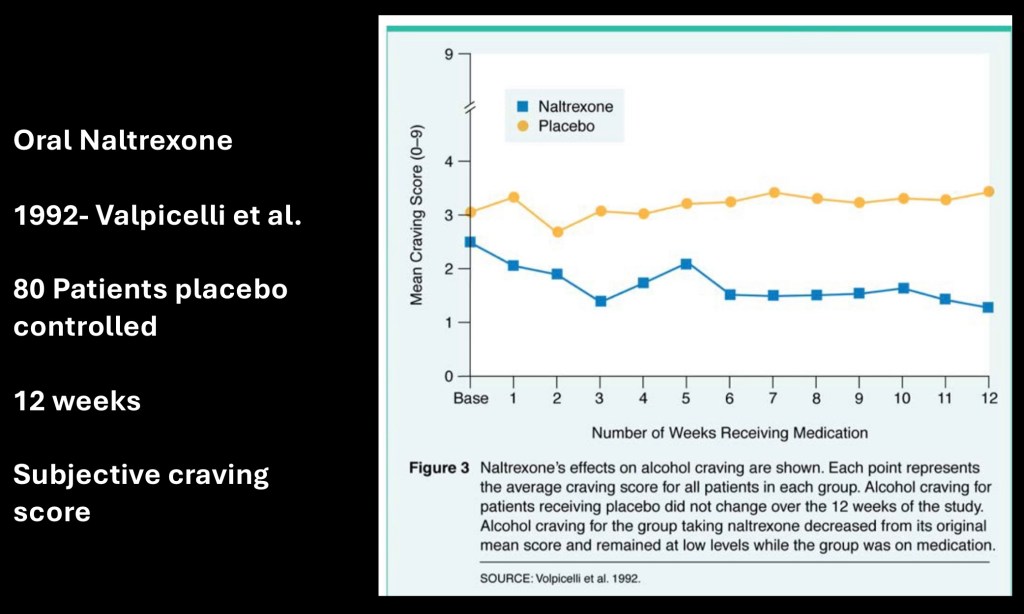
This is from a placebo controlled study of 80 subjects performed by the same group. This graph demonstrates subjective craving score for the 12 week study period. Significantly less craving was experienced by the naltrexone group.

This reflects percent of subjects remaining abstinent during the study period of 12 weeks. By the end of the study around 80% had no relapses as opposed to about 40% in the placebo group.
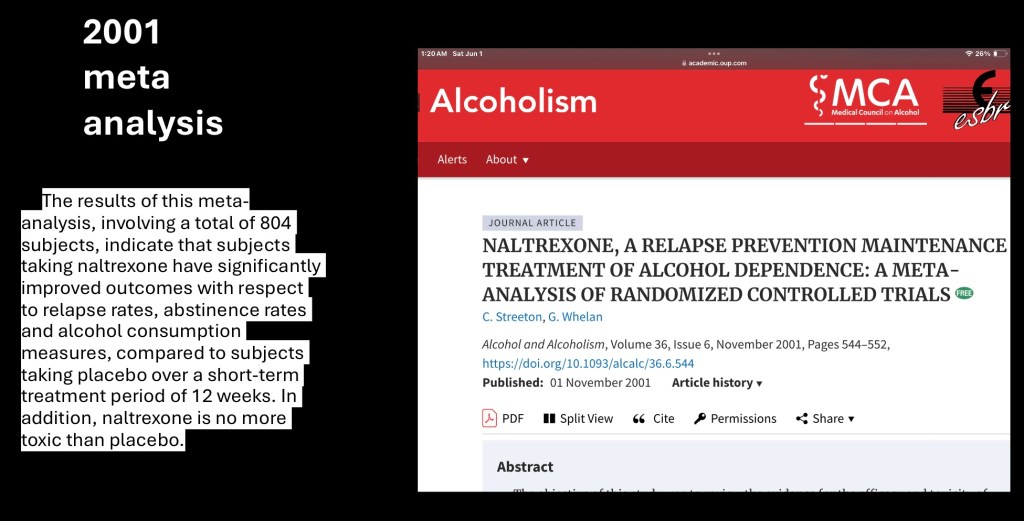
There have been multiple clinical studies on naltrexone to treat AUD usually combined with psychological therapy / peer support group involvement. This meta analysis with a pooled population size of 804 subjects verified significantly improved outcomes with fewer relapses, less alcohol consumed, and greater abstinence rates in naltrexone treatment vs placebo. Effect size is described as modest.

Additional applications for naltrexone have been found or are under investigation,
Bupropion (Wellbutrin) is a drug which has been used for treatment of depression and as an aid in tobacco smoking cessation. It has been considered as a short term weight loss agent although results for bupropion alone have been limited.
Combined with a low dose of naltrexone (8mg) effective weight loss has been demonstrated. It is marketed as Contrave as a pill to be taken daily for short term weight loss.
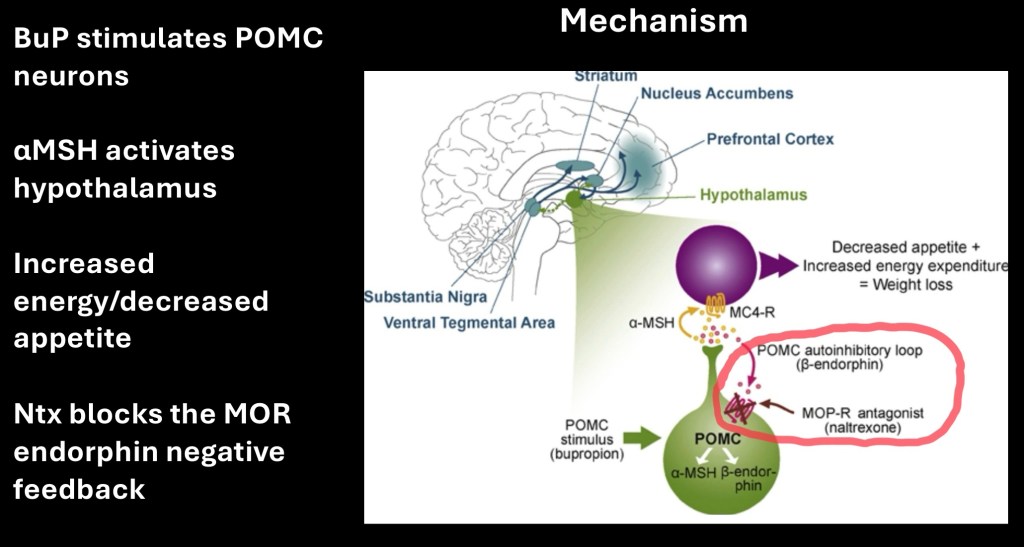
Mechanism of action for BuP Ntx combination. POMC (pro-opriomelanocortin) neurons in the hypothalamus are stimulated by bupropion to release α-MSH (alpha melanocyte stimulating hormone) which causes decreased appetite and increased energy expenditure.
At the same time POMC neurons stimulate production of beta-endorphin. This binds to opioid receptors resulting in a negative feedback mechanism to counter the weight loss effect. Addition of naltrexone blocks the negative feedback resulting in larger weight loss effects
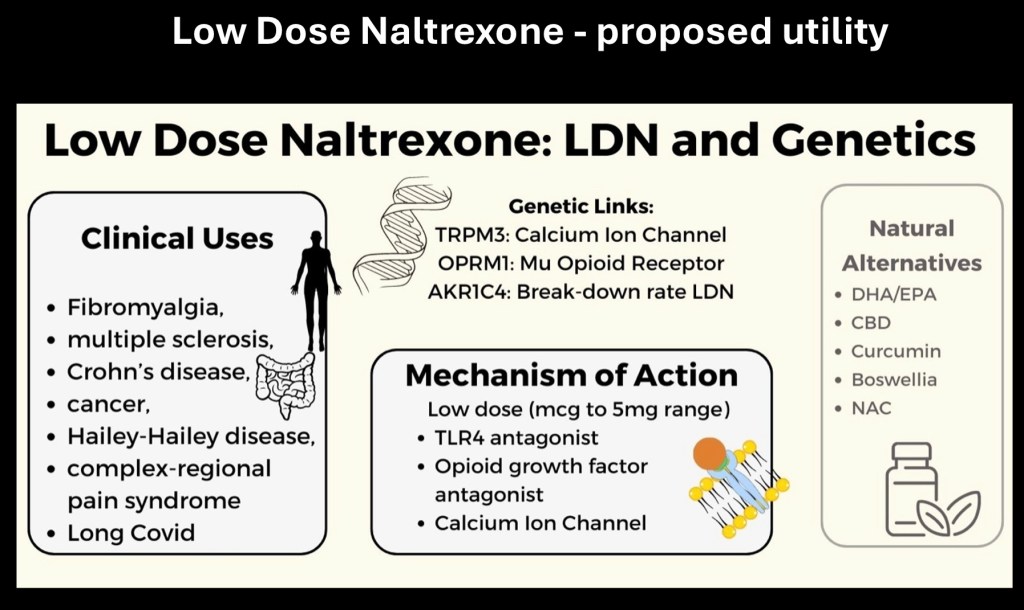
Low dose naltrexone in the 1-5mg range has been more recently studied for novel therapeutic activity in a number of conditions. Naltrexone is thought to have different effects when present in lower doses. It appears to effect modulation of immune responses, activation of genes resulting in anti-cancer properties, and decreased inflammation.
Multiple sclerosis, Crohn’s disease, certain cancers, and fibromyalgia are potential targets.

At low doses a number of reactions are thought to be modulated by naltrexone as summarized above. Larger scale studies are needed for further investigation and indications. Due to its high tolerability and low toxicity it is a promising therapeutic agent.
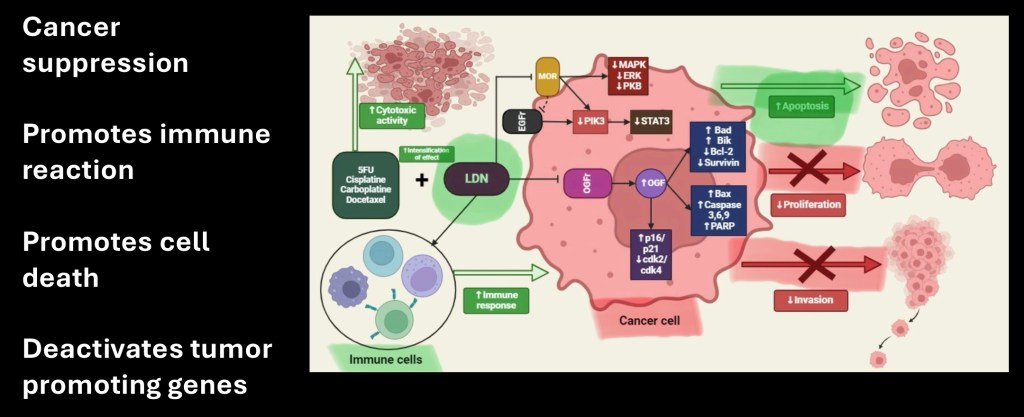
Potential use as an adjunct medication in cancer suppression is under investigation. Possible mechanisms are complex. Key sites of activity are outlined in green above. These include activation of immune cells and activation of genes involved in tumor cell death, prevention of cell proliferation and prevention of invasive properties.
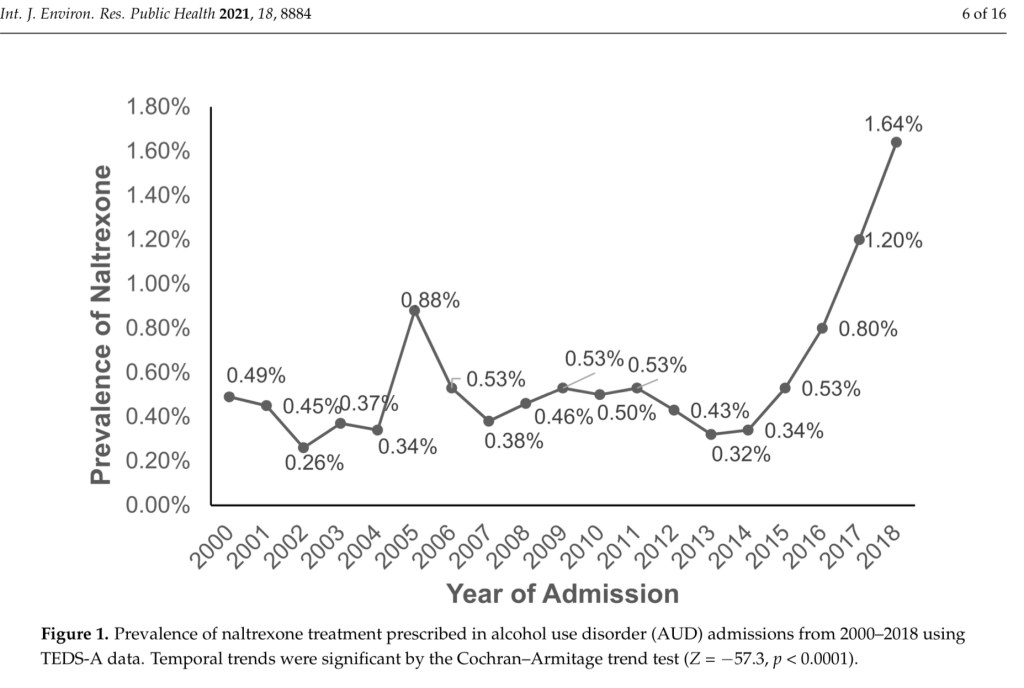
Percentage of hospital patients admitted for AUD receiving Naltrexone prescriptions 2000-2018. While this does not reflect all naltrexone prescriptions these would be a high risk group who may benefit from medical treatment. Only a small minority, 1.6% at most, received naltrexone treatment.
……………………………………………………………………………
The availability and use of pharmacotherapy as an aid in the treatment of substance use disorders is limited at this time. The concept of addiction as a treatable disease in which the medical profession can play a key role still faces significant hurdles. Educating medical professionals, treatment facilities, and the public about this treatment option remains a major hurdle. One such effort, the availability of Naloxone for emergency rescue in opioid overdose is already seeing positive trends in lowering death rates.
Long acting injectable formulations available for treatment of opioid and alcohol use disorders have been shown to improve outcomes for these conditions. Developing selection criteria for this and other treatment alternatives can further decrease morbidity and mortality due to substance use disorders.
One barrier in opioid treatment is the necessity for a 1-2 week pause before initiating antagonist therapy. Referenced here is one strategy to overcome this problem using a tapered approach. Liver disease is a contraindication and a barrier in this population.

Thank you for your time in reviewing this post. Comments and suggestions are always welcome.
Images and data obtained from sources freely available on the World Wide Web. For information and educational purposes only. This post should not be considered medical or professional advice.
References
Naltrexone
Opioid Detoxification and Naltrexone Induction Strategies:
Recommendations for Clinical Practice
Stacey C. Sigmon, Ph.D.1
, Adam Bisaga, M.D.2
Am J Drug Alcohol Abuse. 2012 May ; 38(3): 187–199. doi:10.3109/00952990.2011.653426.
……………………………………………………………………..
Low μ-Opioid Receptor Status in Alcohol Dependence Identified by Combined Positron Emission Tomography and Post-Mortem Brain Analysis
Derik Hermann, Natalie Hirth, Matthias Reimold, Anil Batra,
Neuropsychophamacology 42, pages 606–614 (2017)
……………………………………………………………….
Naltrexone: Not Just for Opioids Anymore
Daniel Sudakin1
Published online: 6 November 2015
# American College of Medical Toxicology 2015
J. Med. Toxicol. (2016) 12:71–75 DOI 10.1007/s13181-015-0512-x
……………………………………………………………….j
, Journal of Lancaster General Health – Journal of Lancaster General Hospital
……………………………………………………………….
Effect of Extended-Release Naltrexone (XR-NTX) on Quality of Life in Alcohol-Dependent Patients
Helen M. Pettinati, PhDa, David R. Gastfriend, MDb,
Alcohol Clin Exp Res. 2009 February ; 33(2): 350–356. doi:10.1111/j.1530-0277.2008.00843.
…………………………………………………………….
Naltrexone in the Treatment of Alcohol Dependence
Authors: John H. Krystal, M.D., Joyce A. Cramer, B.S., William F. Krol, Ph.D.,
Published December 13, 2001
N Engl J Med 2001;345:1734-1739
DOI: 10.1056/NEJMoa011127
Naltrexone in the Treatment of Alcohol Dependence | New England Journal of Medicine
……………………………………………………………..
Low-Dose Naltrexone (LDN)—Review of Therapeutic Utilization
by Karlo Toljan 1,*
Department of Pathophysiology, University of Zagreb School of Medicine, Kispaticeva 12, 10 000 Zagreb, Croatia
Section of Pain Medicine, Department of Anesthesiology, Dartmouth-Hitchcock Medical Center, 1 Medical Center Dr, Lebanon, NH 03756, USA
Department of Anesthesiology, Geisel School of Medicine at Dartmouth, Hanover, NH 03756, USA
Author to whom correspondence should be addressed.
Med. Sci. 2018, 6(4), 82; https://doi.org/10.3390/medsci6040082
Medical Sciences | Free Full-Text | Low-Dose Naltrexone (LDN)—Review of Therapeutic Utilization
……………………………………………………………..
Evidence about the use of naltrexone and for different ways of using it in the treatment of alcoholism
John David Sinclair
Alcohol and Alcoholism, Volume 36, Issue 1, January 2001, Pages 2–10,
…………………………………………………………
Behavioral and Neurobiological Mechanisms of Extinction in Pavlovian and Instrumental Learning
Travis P. Todd, Drina Vurbic, and Mark E. Bouton
Neurobiol Learn Mem. 2014 February ; 0: 52–64. doi:10.1016/j.nlm.2013.08.012.
………………………………………………………………..
Minozzi S, Amato L, Vecchi S, Davoli M, Kirchmayer U, Verster A.
Oral naltrexone maintenance treatment for opioid dependence.
Cochrane Database of Systematic Reviews 2011, Issue 4. Art. No.: CD00133
Naltrexone treatment for opioid dependence: Does its effectiveness depend on testing the blockade?
Maria A. Sullivan,
Drug Alcohol Depend. 2013 November 1; 133(1): 80–85. doi:10.1016/j.drugalcdep.2013.05.030.
………………………………………………
Reductions in Alcohol Craving Following Naltrexone Treatment for Heavy Drinking
Amy W. Helstrom, Frederick C. Blow, Valerie Slaymaker, Henry R. Kranzler, Shirley Leong, David Oslin
Alcohol and Alcoholism, Volume 51, Issue 5, September 2016, Pages 562–566, https://doi.org/10.1093/alcalc/agw038
…………………………………………………
A MULTICENTRE, RANDOMIZED, DOUBLE-BLIND, PLACEBO-CONTROLLED TRIAL OF NALTREXONE IN THE TREATMENT OF ALCOHOL DEPENDENCE OR ABUSE
Jonathan Chick, Raymond Anton, Ken Checinski, Robert Croop, D. Colin Drummond, Roger Farmer, Dominic Labriola, Jane Marshall, Joanna Moncrieff, Marsha Y. Morgan … Show more
Alcohol and Alcoholism, Volume 35, Issue 6, November 2000, Pages 587–593,
……………………………………………………
…………………………………………………………..
Sublingual Buprenorphine-Naloxone Compared With Injection Naltrexone for Opioid Use Disorder: Potential Utility of Patient Characteristics in Guiding Choice of Treatment
Edward V. Nunes Jr., M.D., Jennifer M. Scodes, M.S., Martina Pavlicova, Ph.D., Joshua D. Lee, M.D., M.Sc., Patricia Novo, M.P.A., M.P.H., Aimee N.C. Campbell, Ph.D., John Rotrosen, M.D.
………………………………………………………j
Opioid Detoxification and Naltrexone Induction Strategies:
Recommendations for Clinical Practice
Stacey C. Sigmon, Ph.D.1 Adam Bisaga, M.D.2 Edward V. Nunes, M.D.2 Patrick G. O’Connor, M.D., M.P.H.3 , Thomas Kosten, M.D.4, and George Woody, M.D.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4331107/pdf/nihms661048.pdfi
………………………………………………………….
Naltrexone Hydrochloride | C20H24ClNO4 | CID 5485201 – PubChem
………………………………………………………………
Structure-Activity Relationships of (+)-Naltrexone-Inspired Toll-like Receptor 4 (TLR4) Antagonists.
Selfridge BR kWang X
Journal of Medicinal Chemistry, 05 Jun 2015, 58(12):5038-5052
https://doi.org/10.1021/acs.jmedchem.5b00426 PMID: 26010811 PMCID: PMC4634939
Immunometabolic Modulatory Role of Naltrexone in BV-2 Microglia Cells
by Natalia Kučić 1,* Valentino Rački 2, Roberta Šverko 3, Toni Vidović 3, Irena Grahovac 4 and Jasenka Mršić-Pelčić 5
Int. J. Mol. Sci. 2021, 22(16), 8429; https://doi.org/10.3390/ijms22168429
IJMS | Free Full-Text | Immunometabolic Modulatory Role of Naltrexone in BV-2 Microglia Cells
…………………………………………………………..,..,.,.,
The Role of Toll-Like Receptor 4 in Infectious and Noninfectious Inflammation
Monica Molteni, Sabrina Gemma, and Carlo Rossetti
Dipartimento di Biotecnologie e Scienze della Vita, Universita` degli Studi dell’Insubria,
Mediators of Inflammation
Volume 2016, Article ID 6978936, 9 pages http://dx.doi.org/10.1155/2016/6978936
Non-stereoselective reversal of neuropathic pain by naloxone and
naltrexone:
involvement of toll-like receptor 4 (TLR4)
Mark R. Hutchinson1,2,
………………………………………………………………
Low-Dose Naltrexone as an Adjuvant in Combined Anticancer Therapy
by Marianna Ciwun *, Anna Tankiewicz-Kwedlo and Dariusz Pawlak
Department of Pharmacodynamics, Medical University of Bialystok, Mickiewicza 2C, 15-222 Bialystok, Poland
Author to whom correspondence should be addressed.
Cancers 2024, 16(6), 1240; https://doi.org/10.3390/cancers16061240
Cancers | Free Full-Text | Low-Dose Naltrexone as an Adjuvant in Combined Anticancer Therapy
The efficacy and safety of the naltrexone/bupropion combination for the treatment of obesity: An update
- July 2015Hormones (Athens, Greece) 14(3)
- July 201514(3)
…………………………………………………………….
Safety and efficacy of naltrexone for weight loss in adult patients – a systematic review
Agnieszka Kulak-Bejda1, Grzegorz Bejda2, Napoleon Waszkiewicz1
Arch Med Sci 2021; 17 (4): 940–953
DOI: https://doi.org/10.5114/aoms.2020.96908
……………………………………………………………
May 2015
Naltrexone vs Placebo for the Treatment of Alcohol Dependence
A Randomized Clinical Trial
David W. Oslin, MD1,2; Shirley H. Leong, PhD2; Kevin G. Lynch, PhD1; et al
JAMA Psychiatry. 2015;72(5):430-437. doi:10.1001/jamapsychiatry.2014.3053
https://jamanetwork.com/journals/jamapsychiatry/fullarticle/2174542
NALTREXONE, A RELAPSE PREVENTION MAINTENANCE TREATMENT OF ALCOHOL DEPENDENCE: A META-ANALYSIS OF RANDOMIZED CONTROLLED TRIALS
C. Streeton, G. Whelan
Alcohol and Alcoholism, Volume 36, Issue 6, November 2001, Pages 544–552,
…………………………………………………….
Clinical and biological moderators of response to naltrexone in alcohol dependence: a systematic review of the evidence
James C. Garbutt1, Amy M. Greenblatt2, Suzanne L. West2, Laura C. Morgan2,
………………………………………………………..
A Review of Samidorphan: A Novel Opioid Antagonist
Amna Mohyud Din Chaudhary 1 , Manal F. Khan 2 , Sukhbir S. Dhillon 3 , Sadiq Naveed 3
…………………………………………………………..
Ultra-Low-Dose naloxone or naltrexone to Improve Opioid Analgesia: The History, the Mystery and a novel Approach
Lindsay H. Burns1 and Hoau-Yan wang2
1Pain Therapeutics Inc., San Mateo, CA, USA. 2Deparment of Pharmacology and Physiology, CUNY Medical Scho
Clinical Medicine Insights: Therapeutics 2010:2 857–868
doi: 10.4137/CMT.S4870
https://journals.sagepub.com/doi/pdf/10.4137/CMT.S4870
…………………………………………………….
Naltrexone Inhibits IL-6 and TNFα Production in Human Immune Cell Subsets following Stimulation with Ligands for Intracellular Toll-Like Receptors
Front. Immunol., 10 July 2017
Sec. Antigen Presenting Cell Biology
Volume 8 – 2017 | https://doi.org/10.3389/fimmu.2017.00809
Antagonism of the Met5-enkephalin-opioid growth factor receptor-signaling axis promotes MSC to differentiate into osteoblasts
Nikhil A Thakur 1 , Sean D DeBoyace 1 , Bryan S Margulie
J Orthop Res. 2016 Jul;34(7):1195-205. doi: 10.1002/jor.23135. Epub 2015 Dec 31.
Naltrexone extended-release injection: an option for the management of opioid abuse
Substance abuse and rehabilitation
Robert Taylor Jr1 Robert B Raffa2
Joseph v Pergolizzi Jr
………………………………………………………..
Naltrexone: A History and Future Directions
By A. Benjamin Srivastava, M.D. and Mark S. Gold, M.D.
………………………………………………………….
Injectable extended-release naltrexone for opioid dependence: a double-blind, placebo-controlled, multicentre randomised trial
Evgeny Krupitsky, Edward V Nunes, Walter Ling, Ari Illeperuma, David R Gastfriend, Bernard L Silverman
Lancet 2011; 377: 1506–1
……………………………………………………………
Trends in the Use of Naltrexone for Addiction Treatment among Alcohol Use Disorder Admissions in U.S. Substance Use Treatment Facilities. Int. J. Environ. Res. Public Health 2021, 18, 8884. https://doi.org/10.3390/ ijerph18168884
…………………………………………………………..
Jeff K. 6/24


Leave a comment